Baby You Can Drive My Nanocar
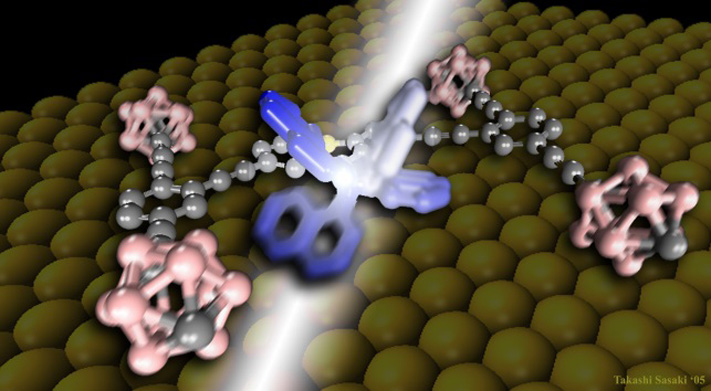

It turns out that off-road driving can be challenging if you’re in a nanocar. A recent research collaboration between NC State and Rice University shows that nanocars – single molecule vehicles with four groups of adamantane serving as wheels – slow down over time when exposed to open air.
Why is this important? Because single molecule vehicles, like nanocars, have the potential to become nanoscopic tools – delivery vehicles at the molecular level for small molecule drugs, proteins or genes. Therefore, learning not only how to control their movement, but also how they perform in ambient, or real-world conditions, is necessary.
Currently, if you want to take your nanocar for a spin the best road conditions are an ultracold vacuum. But Rice chemist James Tour and collaborators including NC State chemist Gufeng Wang wanted to see how the vehicle moved while at room temperature and exposed to the air.
They put nanocars with hydrophobic, or water repelling, wheels on two different glass surfaces: the first had been treated with hydrogen peroxide, which is hydrophilic; the second was further coated with a “nonsticking” polymer. When the hydrophobic wheels hit the hydrophilic glass, the nanocar moves because of thermal energy – energy that comes from the temperature of matter.
Both surfaces were kept at room temperature and the researchers traced the nanocars’ movements for up to 72 hours. During that period, molecules from the open air deposited on the glass surface, causing obstructions that forced the nanocars to slow down, sometimes trapping them completely. The polymer coated glass worked better in terms of keeping the cars moving longer because it kept the “dirty” molecules from the air off the surface. But in both cases, the bumps in the road caused problems.
“The overall project is aimed at improving our capability in controlling the motion of small objects up to single molecule level,” says Wang. “Eventually we’d like to be able to drive these nanocars using an external source, like light. This work is a step toward this goal as we’ve seen for the first time how nanocars’ movements change over time at ambient conditions.”
“Our long-term goal is to make nanomachines that operate in ambient environments,” says Tour. “That’s when they will show potential to become useful tools for medicine and bottom-up manufacturing.”
The research was published in the Journal of Physical Chemistry C, and was supported by the National Science Foundation. Co-authors of the paper are graduate students Victor Garcia-López and Pin-Lei Chu of Rice and graduate students Fang Chen and Tao Jin and postdoctoral scholar Bhanu Neupane of NC State.
This post was originally published in NC State News.


